Chapter 35 - An Overview of the Endocrine System.Section 10 - Endocrine System Toggle Dropdown.Chapter 33 - Basic Structure and Function of the Nervous System.Section 9 - Nervous System Toggle Dropdown.Chapter 32 - The Urinary System and Homeostasis.Chapter 31 - Gross Anatomy of the Kidney.Section 8 - Urinary System Toggle Dropdown.Chapter 29 - Organs and Structures of the Respiratory System.Section 7 - Respiratory System Toggle Dropdown.Chapter 26 - The Adaptive Immune Response: B-lymphocytes and Antibodies.Chapter 25 - The Adaptive Immune Response: T-lymphocytes and Their Functional Types.Chapter 24 - Barrier Defenses and the Innate Immune Response.Section 6 - Circulatory-Immune System Toggle Dropdown.Chapter 20 - Chemical Digestion and Absorption.Chapter 19 - Accessory Organs in Digestion.Chapter 18 - The Small and Large Intestines.Chapter 16 - Digestive System Processes and Regulation.Section 5 - Digestive System Toggle Dropdown.
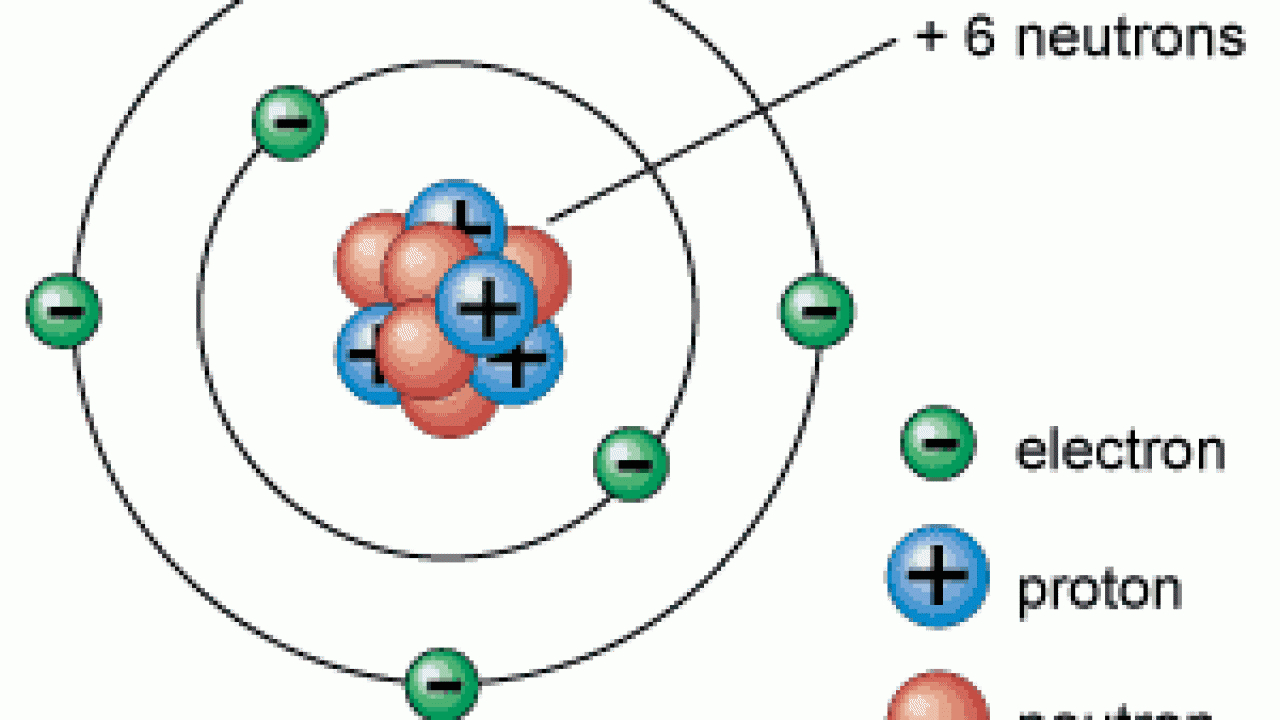
#PARTS OF AN ATOM SKIN#
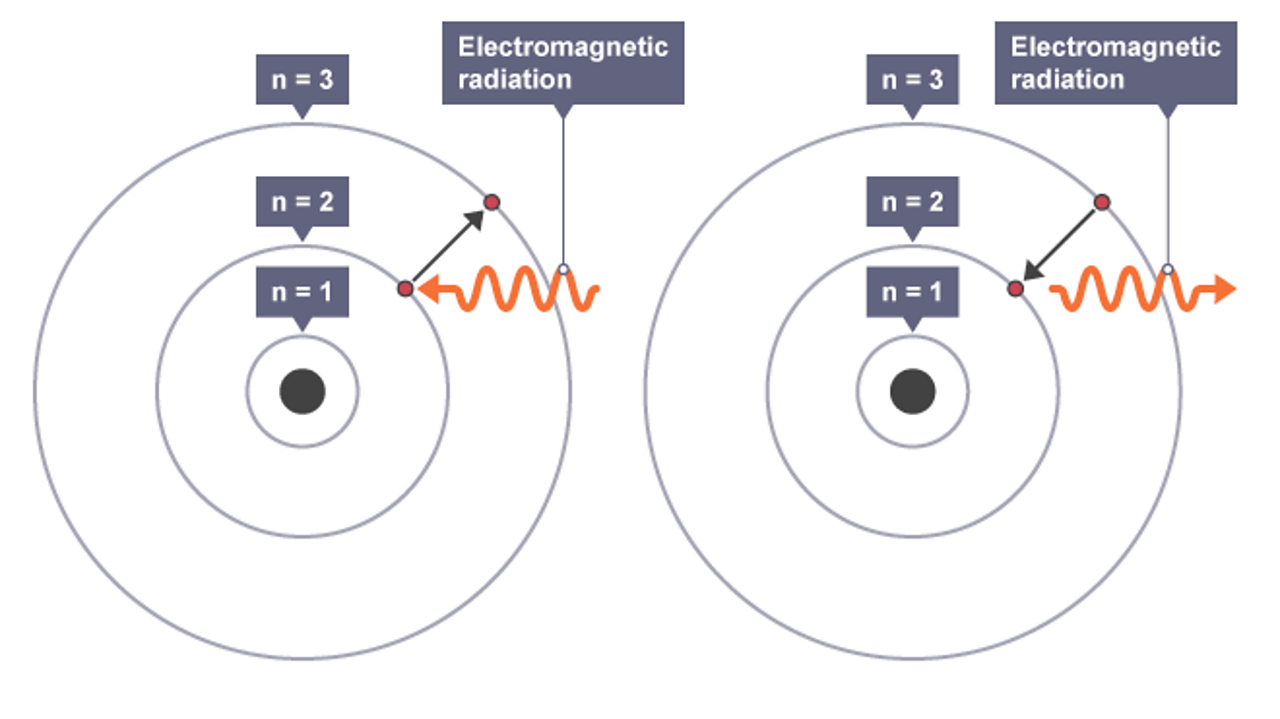
Chapter 15 - Accessory Structures of the Skin.Section 4 - Integumentary System Toggle Dropdown.Chapter 13 - Connective Tissue Supports and Protects.Chapter 12 - Nervous Tissue Mediates Perception and Response.Chapter 4 - Inorganic Compunds Essential to Human Functioning.Chapter 3 - Organic Compounds Essential to Human Functioning.Thumbnail: Ionization energies superimposed on a periodic table. 2.S: Elements, Atoms, and the Periodic Table (Summary) To ensure that you understand the material in this chapter, you should review the meanings of the following bold terms and ask yourself how they relate to the topics in the chapter.2.E: Elements, Atoms, and the Periodic Table (Exercises) These are homework exercises to accompany Chapter 2 of the Ball et al.Some characteristics of the elements are related to their position on the periodic table. 2.8: The Periodic Table The chemical elements are arranged in a chart called the periodic table.2.7: Arrangements of Electrons Electrons are organized into shells and subshells about the nucleus of an atom.2.6: Atomic Masses Atoms have a mass that is based largely on the number of protons and neutrons in their nucleus.Isotopes are atoms of the same element that have different masses. 2.5: Nuclei of Atoms Elements can be identified by their atomic number and mass number.Protons and neutrons are grouped together in the nucleus of an atom, while electrons orbit about the nucleus.


Just as a language has an alphabet from which words are built, chemistry has an alphabet from which matter is described.
